|
Note! - the real force acting on the wall - or an other obstruction in the wind - is in general more complicated to calculate due to drag, turbulence and other effects. The force acting directly on a wall with area 10 m 2 can be calculated as The dynamic pressure in a hurricane with air temperature 20 o C, density of air 1.2 kg/m 3 and wind speed 37 m/s can be calculated as = 12.5 kPa Example - Hurricane and Force acting on a Wall When this occurs, we say the water is under tension. It is possible for the absolute pressure to be negative. 
The dynamic pressure in water with - temperature 20 o C - density 1000 kg/m 3 and velocity 5 m/s - can be calculated as As the pump pushes more liquid through the system, that liquid will overflow through the vapor region and fall down, joining the water in the outlet pipe. By measuring the dynamic pressure in flight, a pitot-static tube (Prandtl tube) can be used to determine the airspeed of an aircraft.Some common densities at atmospheric pressure:Įxample - The Dynamic Pressure in a Water Flow As we have seen, dynamic pressure appears in Bernoulli’s equation even though that relationship was originally derived using energy conservation. The dynamic pressure is therefore used in the definition of the lift coefficient and the drag coefficient. In particular, the aerodynamic forces acting on an object as it moves through the air are directly proportional to the dynamic pressure. We have performed this simple derivation to determine the form of the dynamic pressure, but we can use and apply the idea of dynamic pressure in much more complex flows, like compressible flows or viscous flows. The dynamic pressure is a defined property of a moving flow of gas. Each term in this equation has the dimensions of a pressure (force/area) ps is the static pressure, the constant pt is called the total pressure, and is called the dynamic pressure because it is a pressure term associated with the velocity u of the flow.5 * r * u^2ĭynamic pressure is often assigned the letter q in aerodynamics: q =. This equation looks exactly like the incompressible form of Bernoulli’s equation. Integrating this differential equation: ps +. This phenomenon is called Pascal’s principle. Remarkably, the pressure is transmitted undiminished. Performing a little algebra: dp/dx + r * u * du/dx = 0įor a constant density (incompressible flow) we can take the “r * u” term inside the differential: dp/dx + d(.5 * r * u^2)/dx = 0Īnd then gather all of the terms: d(p +. Since atoms in a fluid are free to move about in an enclosed fluid, they transmit the pressure to all parts of the fluid and to the walls of the container. Where r is the density of the gas, p is the pressure, x is the direction of the flow, and u is the velocity in the x-direction. 
It is crucial, for example, in determining whether an object sinks or floats in a fluid. Density, as you will see, is an important characteristic of substances. Fluid statics is the physics of stationary fluids. 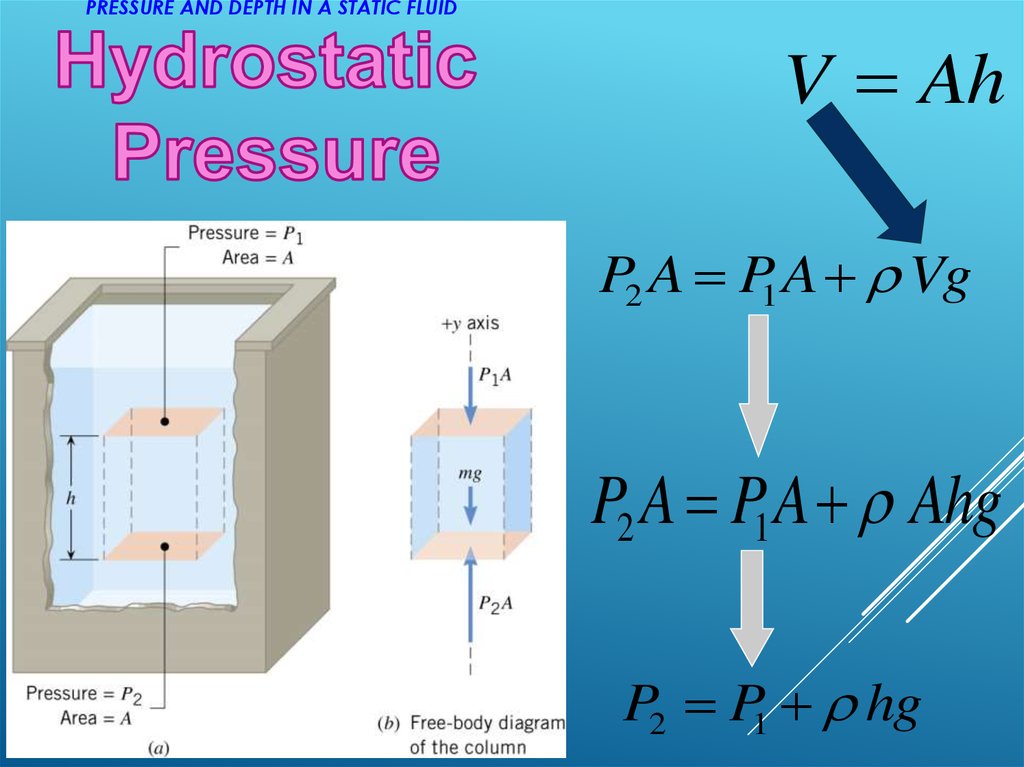
To understand dynamic pressure, we begin with a one-dimensional version of the conservation of linear momentum for a fluid. A fluid is a state of matter that yields to sideways or shearing forces. This leads to the definition of dynamic pressure. But if the gas is moving, the measured pressure depends on the direction of motion. If gas is static and not flowing, the measured pressure is the same in all directions. Although pressure itself is a scalar quantity, we can define a pressure force to be equal to the pressure (force/area) times the surface area in a direction perpendicular to the surface. The change in pressure during any process is governed by the laws of thermodynamics. On the large scale, the pressure of a gas is a state variable, like the temperature and the density. The pressure of a gas is a measure of the average linear momentum of the moving molecules of a gas. The change in momentum produces a force on the walls which is related to the gas pressure. During collisions with the walls, there is a change in velocity and therefore a change in the momentum of the molecules. The molecules of a gas are in constant, random motion and frequently collide with each other and with the walls of any container. On the small scale, from the kinetic theory of gases, a gas is composed of a large number of molecules that are very small relative to the distance between molecules. There are two ways to look at pressure: (1) the small-scale action of individual air molecules or (2) the large-scale action of a large number of molecules. Because understanding what pressure is and how it works is so fundamental to the understanding of aerodynamics and propulsion, we are including several slides on pressure in the Beginner’s Guide. An important property of any gas is its pressure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
